Describe How Red Blood Cells Use Osmosis
In a similar procedure using plant epidermis the student observed that. Red blood cell glucose transporters GLUT-1 are regulated by intracellular ATP and AMP levels.
A student wanted to observe some red blood cells under a microscope.

. When blood is centrifuged the heaviest blood components moving to the bottom and the lightest ones moving to the top. Examine the mount under low medium and high power. A solution will be hypertonic to a cell if its solute concentration is higher.
Add a drop or two of 3 sodium chloride to the edge of the cover slip. By placing red blood cells in solutions of differing osmolarities and tonicities this experiment demonstrates the effects of osmosis and the resultant changes in cell volume. A concept called osmosis where water for example moves through a semipermeable membrane into a solution of a higher solute concentration that tends to equalise concentrations of solute on the inside and outside of the membrane explains why Baxters cells lacked water.
Baxters red blood cells began to shrink because water moved outside the cell to reach. It is a circle with a big beveled area in the middle of it. When viewed at high power the student observed that the red blood cells had burst.
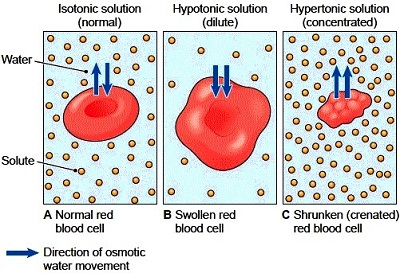
Listed below are more examples of Osmosis. 2 Describe the appearance of red blood cells in a hypotonic solution The red blood cell appears to have balloon out to make a round cell. If abnormal blood cells are placed in a raft with proper lower solute concentration than mercury found against the cells water moves into the cells by osmosis causing the cells to.
Osmosis and Red Blood Cells The Effect Of The Effects Of Osmosis On The Cell. GLUT-1 is one of the major glucose transporters for red blood cells. They placed a small sample of blood onto a microscope slide and added a drop of distilled water.
A A- B B- AB AB- O O-. Osmosis and cells play integral roles in biological life. Define osmosis and describe the effects of placing red blood cells in the following solutions 1 Hypertonic salline solution 3Nacl 2 Hypertonic salline solution 045Nacl 3Isotonicnormal salline 09Nacl.
Using hemoglobin standard solutions where known concentrations of hemoglobin are produced the proportion of hemolysis and the effect of this on resultant hematocrit can be estimated. In an animal cell osmosis helps in absorbing water from the intestines to the blood. The concentration of ions and other solute molecules is higher inside the cell than outside it so water moves into the cell via osmosis.
Glucose travels from the intestinal lumen into the intestinal epithelial cells through active transport and then glucose enters red blood cells through facilitated diffusion. Draw the solution under the cover slip by using the paper towel. Example of Osmosis.
Osmosis has a significant role to play in plants animals and also in humans. What causes red blood cells to burst. Describe what you think happened in the body to result in.
So starting at the bottom of the tube. Describe the appearance of individual red blood cells. A good example of osmosis is seen when red blood cells are placed into fresh water.
This Osmosis High-Yield Note provides an overview of Blood Components and Function essentials. Observe the red blood cells until no further change occurs. The lack of enzyme causes your red blood cells to rupture and die when they come into contact with certain substances in the bloodstream.
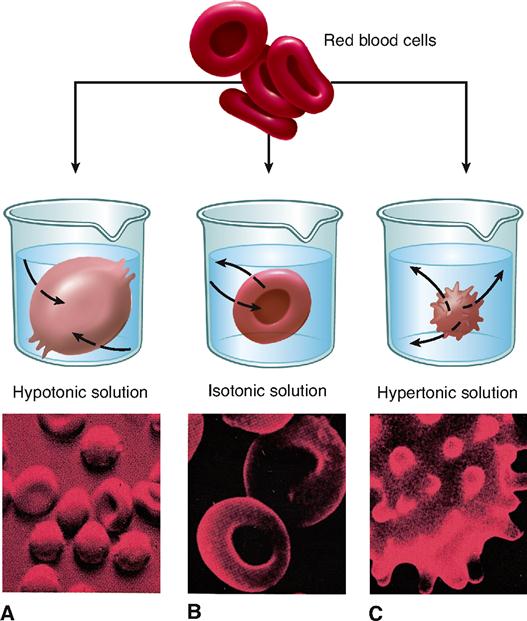
Osmosis in Red Blood Cells In the diagram below Red Blood Cells have been subjected to different conditions that exist in the human body. The erythrocytes or red blood cells at the bottom the buffy coat--which contains platelets and immune cells in the middle and plasma at the top. Resistance Of Movement Of The Cell.
When both classification systems are combined there are eight possible blood types. As far as red blood cells are concerned osmosis is a critical process because it ultimately determines healthiness. Surrounding the cell then osmosis will result by the cell gaining water For example if you The membrane separating the inside of an animal cell from its outside allows water to pass through.
Three termshyerptonic hypotonic and isotonicare used to describe whether a solution will cause water to move into or out of a cell. Osmosis is the traveling of water across a membrane. The absorption of water from the soil is due to osmosis.
If a cell contains a high concentration of salt or other dissolved. What blood cell osmosis lab reports are found in red blood can result in blood cells are mixed with the highest water would you feel dizzy or liver. This movement occurs through osmosis because the cell has more free water than the solution.
If a cell is placed in a hypertonic solution there will be a net flow of water out of the cell and the cell will lose volume. TaylorIn order to regulate osmosis a cell uses a fluid. This causes the cells to swell.
If a cell is to perform its functions it must maintain a steady state. By definition Osmosis is the diffusion of a solvent such as water. Osmosis Describes the diffusion of water molecules across a selectively permeable membrane from an area of higher concentration to an area of lower concentration.
Red blood cells as in all animal cells dont have cell walls. It is important for a cells survival to regulate osmosis in order to maintain an optimal internal environment according to Student Study Guide for Campbells BIOLOGY Fourth Edition by Martha R. Observe under the cell in.
Being that blood cells are selectively permeably decides what is allowednot allowed inside the ways in which water travel in and out of the cells is important. Blood Components Function Figure 437 Blood types are reported as ABO group and Rh or -. The cell membrane of the red blood cells is a semipermeable membrane.
1 Describe the appearance of red blood cells in an isotonic solution The red blood cell in isotonic solution looks like a bowl. Overall three distinct layers form. When placing a red blood cell in any hypertonic solution there will be a movement of free water out of the cell and into the solution.
When red blood cells are placed in pure water water rapidly enters the cells by osmosis and causes the cells to burst a phenomenon known as hemolysis.

7th Grade Science Day To Day March 2013 7th Grade Science Osmosis Medical School Studying

What Will Happen When Red Blood Cells Are Placed In Class 12 Biology Cbse
What Are Diffusion And Osmosis Enotes Com

Pin On Nursing School Study Tips Nclex

Osmosis Animal Cell Stock Vector Illustration Of Shrinkage 94695691
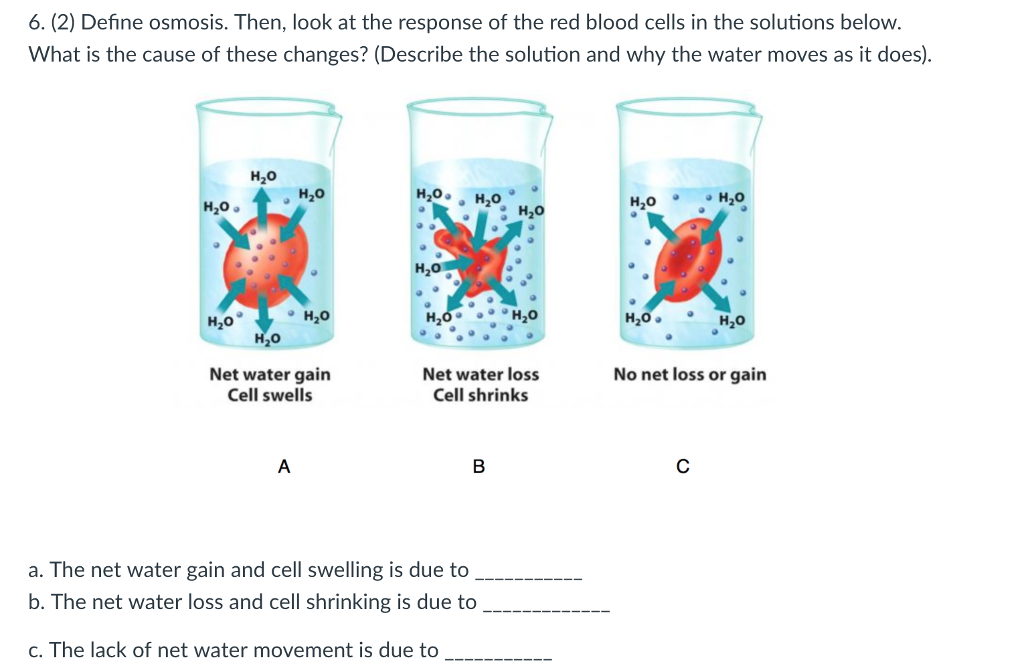
Solved 6 2 Define Osmosis Then Look At The Response Of Chegg Com

Science Source Images Red Blood Cells In Solution Demonstrating Osmosis Three Red Blood Cells In Different Solutions Isotonic Hypertonic And Hypotonic Click On The Following Link For Wall Art Masks And

Tonicity And Osmosis On Red Blood Cell Stock Vector Illustration Of Osmotic Desalination 102742008

Stock Image Jf3100 01b475ur Science Source Search Medical Scientific Stock Photos At Medicalimages Com

250 Cell Osmosis Stock Photos Pictures Royalty Free Images Istock
Physiology Of Cells Basicmedical Key

Osmotic Burst Of Blood Cells Youtube
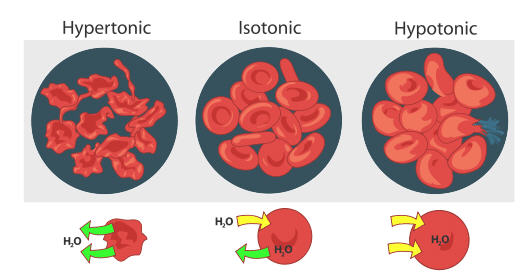
8 4 Osmosis And Diffusion Chemistry Libretexts
Solved Why Does The Osmosis Of Water Initially Cause The Shrinkage Of Red Course Hero

Stock Image Jf3106 01b475ux Science Source Search Medical Scientific Stock Photos At Medicalimages Com

Stock Image Jf3105 01b475uw Science Source Search Medical Scientific Stock Photos At Medicalimages Com

Osmosis In Red Blood Cells Photograph By Science Photo Library

Isotonic Hypotonic And Hypertonic Solutions Effects On Animal Cells Tonicity And Osmosis This Diagram Shows The

One Experiment From The Osmosis Lab A Red Blood Cell Is On The Left Download Scientific Diagram

Comments
Post a Comment